Are you a healthcare
professional?
The information provided in this website is intended for US healthcare professionals only, I confirm that I am a US healthcare professional.
What is Chagas disease?
The FDA defines Chagas disease as a parasitic infection caused by the Trypanosoma cruzi parasite.1 It is usually transmitted by infected insects called triatomine bugs, or kissing bugs.1-3 Kissing bugs, also known as chinches in Mexico and Central America, are found throughout the Americas—from Argentina to the southern United States.3,4
These insects feed at night when their hosts are sleeping.3,4 As they feed, the bugs defecate.3,4 Their feces contain the T cruzi parasite, which can cause Chagas disease when hosts scratch the bite and the feces enter their bloodstream.3,4
Other forms of transmission include3-5
Congenital transmission from an infected mother to a fetus
Blood transfusion
Organ transplantation
Chagas disease is rarely transmitted through contaminated food and drink.3,4 It is not a communicable disease and is not transmitted from person to person.3,4
Chagas disease affects an estimated
300,000 people living in the United States5
Chagas disease is an endemic infection throughout much of Mexico, Central America, and South America.3,4 It has been designated as a neglected tropical disease by the FDA, which considers it an emerging public health concern.1,2
Phases of Chagas disease
Chagas disease has an acute phase and a chronic phase. If left untreated, the infection becomes lifelong.2
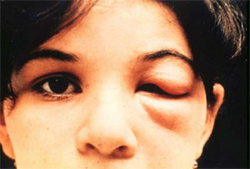
The acute phase occurs immediately after infection and may last up to the first few weeks or months of infection.6
- Patients may develop mild flu-like symptoms, including fever, body aches, and swelling at the bite site, or remain asymptomatic3,4
- The most recognized marker of acute Chagas disease is called Romaña's sign, which includes swelling of the eyelid on the side of the face near the bite wound6
- Although symptoms may resolve, if left untreated the infection persists6
- Additionally, these symptoms are not unique to Chagas disease, which can make recognizing the disease even more challenging6
- In rare cases, severe inflammation of the heart muscle or the brain and lining around the brain can occur7
Most infected people then enter into the chronic phase of the disease, which can go undetected for decades.3 This is called the chronic indeterminate phase because many people remain asymptomatic and are unaware of their infection.8
It is estimated that 20% to 30% of chronically infected people will develop complications.3
- These can include serious gastrointestinal or cardiac diseases, such as an enlarged esophagus or colon, as well as cardiomyopathy, heart failure, stroke, and cardiac arrest5,6
Diagnosing Chagas disease
Chagas disease can only be diagnosed through analysis of a specific blood test to detect the presence of the parasitic infection.5,6 The reference diagnostic laboratory for parasitic diseases at the CDC can offer assistance.5
Learn more about diagnosing Chagas disease at
Treating Chagas disease
Benznidazole Tablets are the first FDA-approved treatment for Chagas disease. Benznidazole Tablets are indicated in pediatric patients 2 to 12 years of age for the treatment of Chagas disease caused by Trypanosoma cruzi. This indication received accelerated approval based on the number of treated patients who were successfully treated in clinical trials.9
Learn more aboutBenznidazole Tablets
For additional information on Chagas disease, please visit
INDICATIONS AND USAGE
Benznidazole Tablets, a nitroimidazole antimicrobial, is indicated in pediatric patients 2 to 12 years of age for the treatment of Chagas disease (American trypanosomiasis) caused by Trypanosoma cruzi.
This indication is approved under accelerated approval based on the number of treated patients who became Immunoglobulin G (IgG) antibody negative against the recombinant antigens of T cruzi. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
IMPORTANT SAFETY INFORMATION
Contraindications
Benznidazole Tablets are contraindicated in patients with a history of hypersensitivity reactions to benznidazole or other nitroimidazole derivatives. Reactions have included severe skin and soft tissue reactions. Benznidazole Tablets is also contraindicated in patients who have taken disulfiram within the last two weeks. Psychotic reactions may occur in patients who are using benznidazole and disulfiram concurrently. Consumption of alcoholic beverages or products containing propylene glycol is contraindicated in patients during and for at least 3 days after therapy with Benznidazole Tablets. A disulfiram-like reaction (abdominal cramps, nausea, vomiting, headaches, and flushing) may occur due to the interaction between alcohol or propylene glycol and benznidazole. Benznidazole Tablets are contraindicated in patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to benznidazole in patients with Cockayne syndrome.
Warnings and Precautions
Potential for Genotoxicity and Carcinogenicity: Genotoxicity has been demonstrated in humans, in vitro in several bacterial species and mammalian cell systems, and in vivo in rodents. A study evaluating the cytogenetic effect of benznidazole in pediatric patients ranging from 11 months to 11 years of age (the safety and effectiveness of Benznidazole Tablets in patients less than 2 years old has not been established) with Chagas disease demonstrated a two-fold increase in chromosomal aberrations. In pediatric patients with Chagas disease who were treated with benznidazole, the median incidence of micronucleated interphase lymphocytes in 20 patients increased two-fold compared to pre-dose values. In the same study, the mean incidence of chromosomal aberrations in 10 patients also increased two-fold compared to pre-dose values.
Carcinogenicity has been observed in mice and rats treated chronically with nitroimidazole agents, which are structurally similar to benznidazole. Similar data have not been reported for benznidazole. It is not known whether benznidazole is associated with carcinogenicity in humans.
Embryo-Fetal Toxicity: Based on animal studies, Benznidazole Tablets can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Pregnancy testing is recommended for women of reproductive potential prior to Benznidazole Tablets therapy. Advise women of reproductive potential to use effective contraception during treatment with Benznidazole Tablets and for 5 days after the last dose.
Hypersensitivity Skin Reactions: Serious skin and subcutaneous disorders, including acute generalized exanthematous pustulosis (AGEP), toxic epidermal necrolysis (TEN), erythema multiforme, and eosinophilic drug reactions, have been reported with benznidazole. Discontinue treatment at first evidence of these serious cutaneous reactions. Extensive skin reactions, such as rashes, have also been reported. Most cases occurred after approximately 10 days of treatment with benznidazole. Most rashes resolved with treatment discontinuation. In case of skin reactions presenting with additional symptoms or signs of systemic involvement, such as lymphadenopathy, fever, and/or purpura, discontinuation of treatment is recommended.
Central and Peripheral Nervous System Effects: Benznidazole Tablets can cause paresthesia or symptoms of peripheral neuropathy that may take several months to resolve. Headache and dizziness have been reported. In cases where neurological symptoms occur, immediate discontinuation of treatment is recommended. In most cases, symptoms occur late in the course of treatment.
Hematological Manifestations of Bone Marrow Depression: There have been reports of hematological manifestations of bone marrow depression, such as neutropenia, thrombocytopenia, anemia, and leukopenia, which resolved after treatment discontinuation. Patients with hematological manifestations of bone marrow depression must take Benznidazole Tablets only under strict medical supervision. Monitor complete blood count. Total and differential leukocyte counts are recommended before, during, and after therapy.
Adverse Reactions
The most common adverse reactions observed were abdominal pain (25%), rash (16%), decreased weight (13%), and headache (7%).
Use in Specific Populations
Lactation Risk: Limited published literature based on breast milk sampling reports that benznidazole is present in human milk. There are no reports of adverse effects on the breastfed infant and no information on the effects of benznidazole on milk production. Because of the potential for serious adverse reactions, and transmission of Chagas disease, advise patients that breastfeeding is not recommended during treatment with Benznidazole Tablets.
Male Infertility: Based on findings in rodents, Benznidazole Tablets may impair fertility in males of reproductive potential. It is not known whether effects on fertility are reversible.
Before prescribing Benznidazole Tablets and for further safety information, please read the full Prescribing Information here.
References: 1. Chagas disease: an emerging public health concern. US Food and Drug Administration Office of Minority Health website. https://www.fda.gov/downloads/ForConsumers/ByAudience/MinorityHealth/UCM470160.pdf. Accessed December 15, 2017. 2. Montgomery SP, Parise ME, Dotson EM, Bialek SR. What do we know about Chagas disease in the United States? Am J Trop Med Hyg. 2016;95(6):1225-1227. 3. Chagas disease: provider fact sheet. Centers for Disease Control and Prevention website. https://www.cdc.gov/parasites/chagas/resources/factsheet.pdf. Accessed December 15, 2017. 4. Chagas disease fact sheet. Centers for Disease Control and Prevention website. https://www.cdc.gov/parasites/chagas/resources/onepage.pdf. Accessed December 27, 2017. 5. Chagas disease in the Americas. Centers for Disease Control and Prevention website. https://www.cdc.gov/parasites/chagas/resources/chagasdiseaseintheamericas2015.pdf. Accessed December 15, 2017. 6. Detailed FAQs. Centers for Disease Control and Prevention website. https://www.cdc.gov/parasites/chagas/gen_info/detailed.html. Accessed December 15, 2017. 7. Parasites–American Trypanosomiasis (also known as Chagas Disease). Disease. Centers for Disease Control and Prevention website. https://www.cdc.gov/parasites/chagas/disease.html. Accessed December 27, 2017. 8. Chagas disease: what U.S. clinicians need to know. Centers for Disease Control and Prevention website. https://www.cdc.gov/parasites/cme/chagas/lesson_2/1.html. Accessed December 15, 2017. 9. FDA approves first U.S. treatment for Chagas disease news release. Silver Springs, MD: US Food and Drug Administration; August 29, 2017. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ ucm573942.htm. Accessed January 12, 2018.
Indications and Usage and Important Safety Information
INDICATIONS AND USAGE
Benznidazole Tablets, a nitroimidazole antimicrobial, is indicated in pediatric patients 2 to 12 years of age for the treatment of Chagas disease (American trypanosomiasis) caused by Trypanosoma cruzi.
This indication is approved under accelerated approval based on the number of treated patients who became Immunoglobulin G (IgG) antibody negative against the recombinant antigens of T cruzi. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
